It is interesting to think that at this very moment all of us, every living terrestrial organism, are living in a state of pressure. We normally don’t feel it the human body is primarily made up of liquid, and liquids are basically non compressible. At times, however, we do notice changes of pressure, primarily in our ears. This is often described as a “pop” and it occurs when our elevation changes, like when we fly or driving in the mountains. This is because our ears have an air space in them, and air, like all other gases, is compressible.
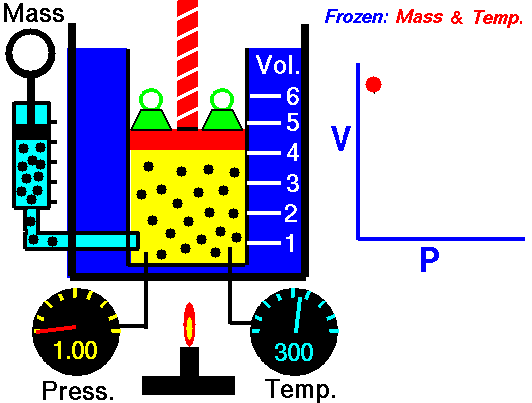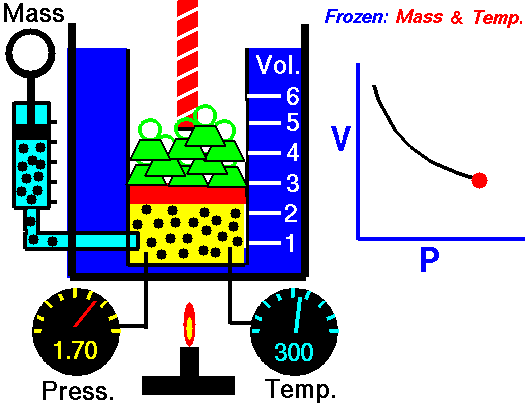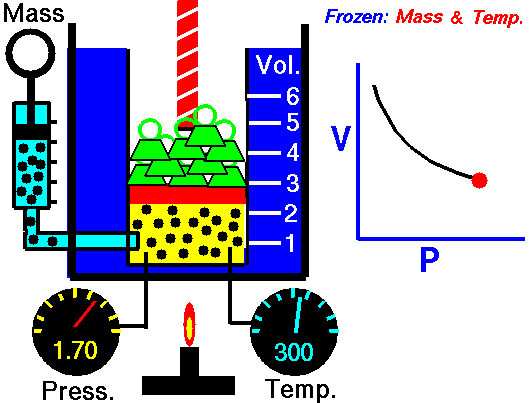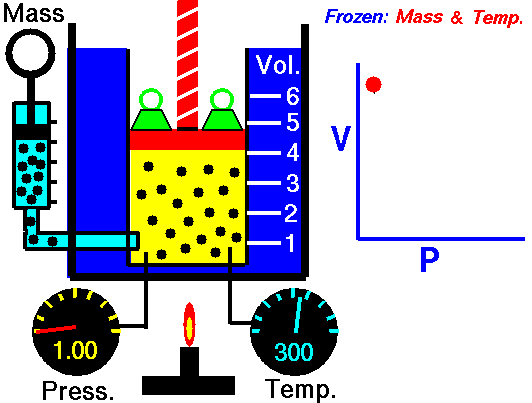
Robert Boyle was one of the first people to study this phenomena in 1662. He formalized his findings into what is now called Boyle’s law, which states that “If the temperature remains constant, the volume of a given mass of gas is inversely proportional to the absolute pressure” Essentially, what Boyle was saying is that an ideal gas will compress proportionately to the amount of pressure exerted on it. For example, if you have a 1 cubic meter balloon and double the pressure on it, it will be compressed to ½ a cubic meter. Increase the pressure by 4, and the volume will drop to 1/4 of its original size, and so on.
The law can also be stated in a slightly different manner, that the product of absolute pressure (p) and volume (V) is always constant (k); p x V = k, for short. While Boyle derived the law solely on experimental grounds, the law can also be derived theoretically based on the presumed existence of atoms and molecules and assumptions about motion and that all matter is made up of a large number of small particles (atoms or molecules) all of which are in constant, motion. These rapidly moving particles constantly collide with each other and with the walls of their container (also known as the kinetic theory).
Another example of Boyle’s law in action is in a syringe. In a syringe, the volume of a fixed amount of gas is increased by drawing the handle back, thereby lessening the pressure. The blood in a vein has higher pressure than the gas in the syringe, so it flows into the syringe, equalizing the pressure differential. Boyle’s law is one of three gas laws which describe the behavior of gases under varying temperatures, pressures and volumes. The other two laws are Gay-Lussac’s law and Graham’s law. Together, they form the ideal gas law.
For an animated demonstration of Boyle’s Law, click here.
We have written many articles about Boyle’s Law for Universe Today. Here’s an article about air density, and here’s an article about the Boltzmann Constant.
If you’d like more info on Boyle’s Law, check out NASA’s Boyle’s Law Page, and here’s a link to the Boyle’s Law Lesson.
We’ve also recorded an episode of Astronomy Cast. Listen here, Question Show: The Source of Atmospheres, The Vanishing Moon and A Glow After Sunset.
Sources:
http://en.wikipedia.org/wiki/Boyle%27s_law
http://en.wikipedia.org/wiki/Ideal_gas
http://www.chm.davidson.edu/vce/gaslaws/boyleslaw.html
http://home.flash.net/~table/gasses/boyle1.htm
http://www.wisegeek.com/what-is-boyles-law.htm
http://www.grc.nasa.gov/WWW/K-12/airplane/boyle.html